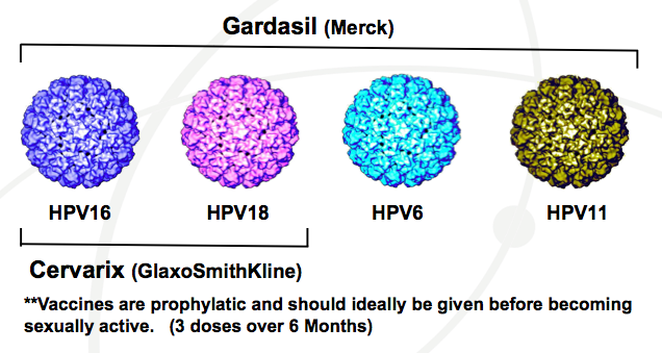
Cervarix and Gardasil
|
Fortunately, there are vaccines available to the public that can protect the human population from these potentially fatal diseases and cancers. The two HPV vaccines that are licensed by the Food and Drug Administration (FDA) are Cervarix and Gardasil. They are both made with the protein outer coat of the actual virus and CDC recommends that they be given in three doses over a 6-month period to preteens before the adolescents get involved in sexual activity. Both vaccines are injected into the body with a harmless form of the virus that does not cause the disease. |

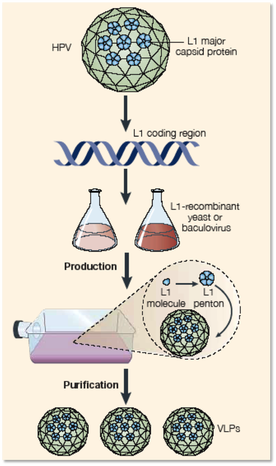
Gardasil is the quadrivalent HPV vaccine that was discovered first and was approved by the FDA in 2006 for females ages 9-26 to prevent cancers. It differs from Cervarix because it expresses the L1 major capsid protein in yeast and is manufactured by Merck & Co. Gardasil protects against HPV types 6, 11, 16, and 18 , while Cervarix has been proven to only protect against types 16 and 18, which makes it a bivalent vaccine rather than a quadrivalent vaccine. Manufactured by GlaxoSmithKline Biologicals, Cervarix expresses the L1 major capsid protein in insect cells rather than yeast. Overall, both vaccines are highly efficient in preventing infections that can be caused from the types of HPV they target.